| Tissue | Expression Dynamics | Abbreviation |
| Colorectum (GSE201348) | 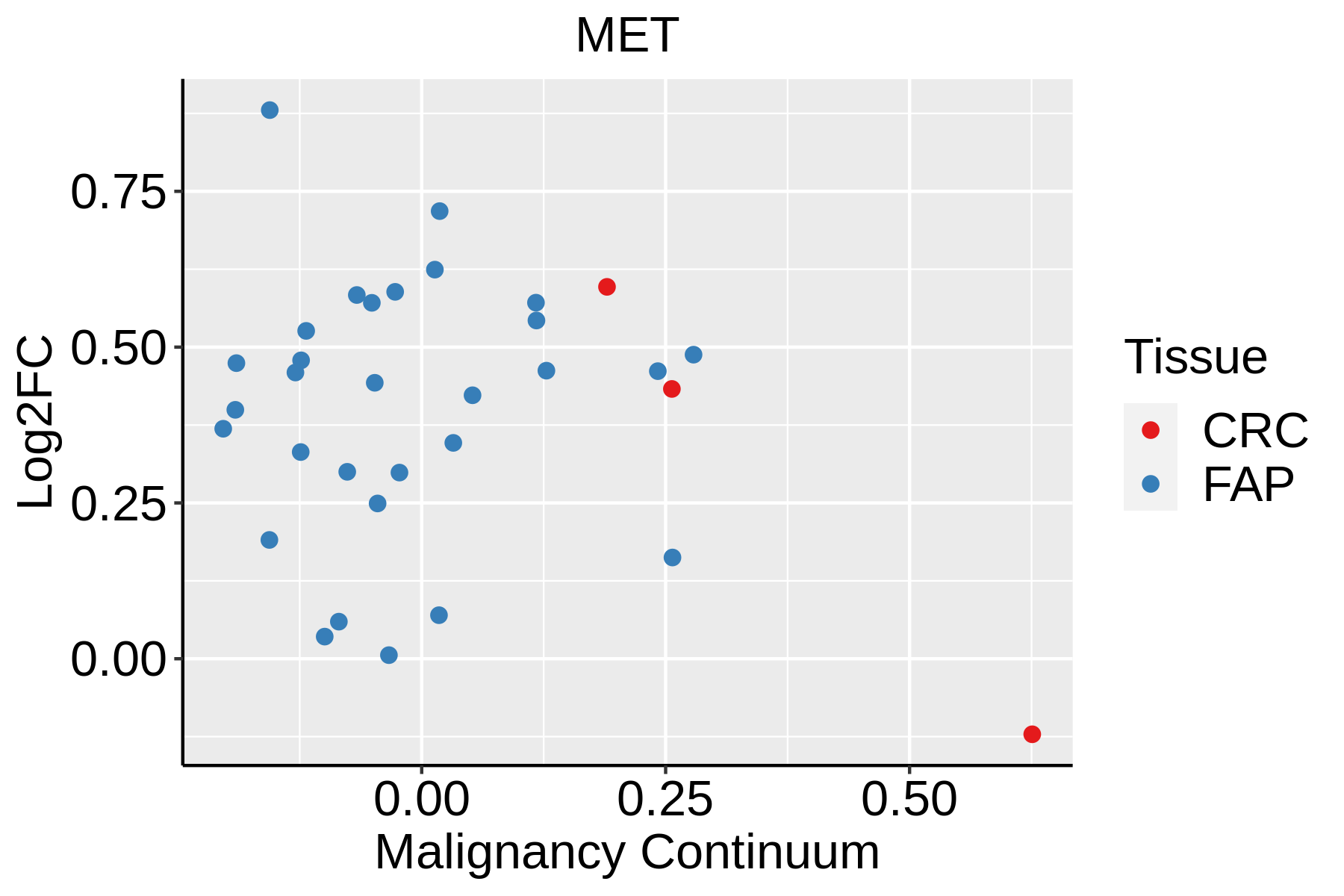 | FAP: Familial adenomatous polyposis |
| CRC: Colorectal cancer |
| Colorectum (HTA11) |  | AD: Adenomas |
| SER: Sessile serrated lesions |
| MSI-H: Microsatellite-high colorectal cancer |
| MSS: Microsatellite stable colorectal cancer |
| Esophagus | 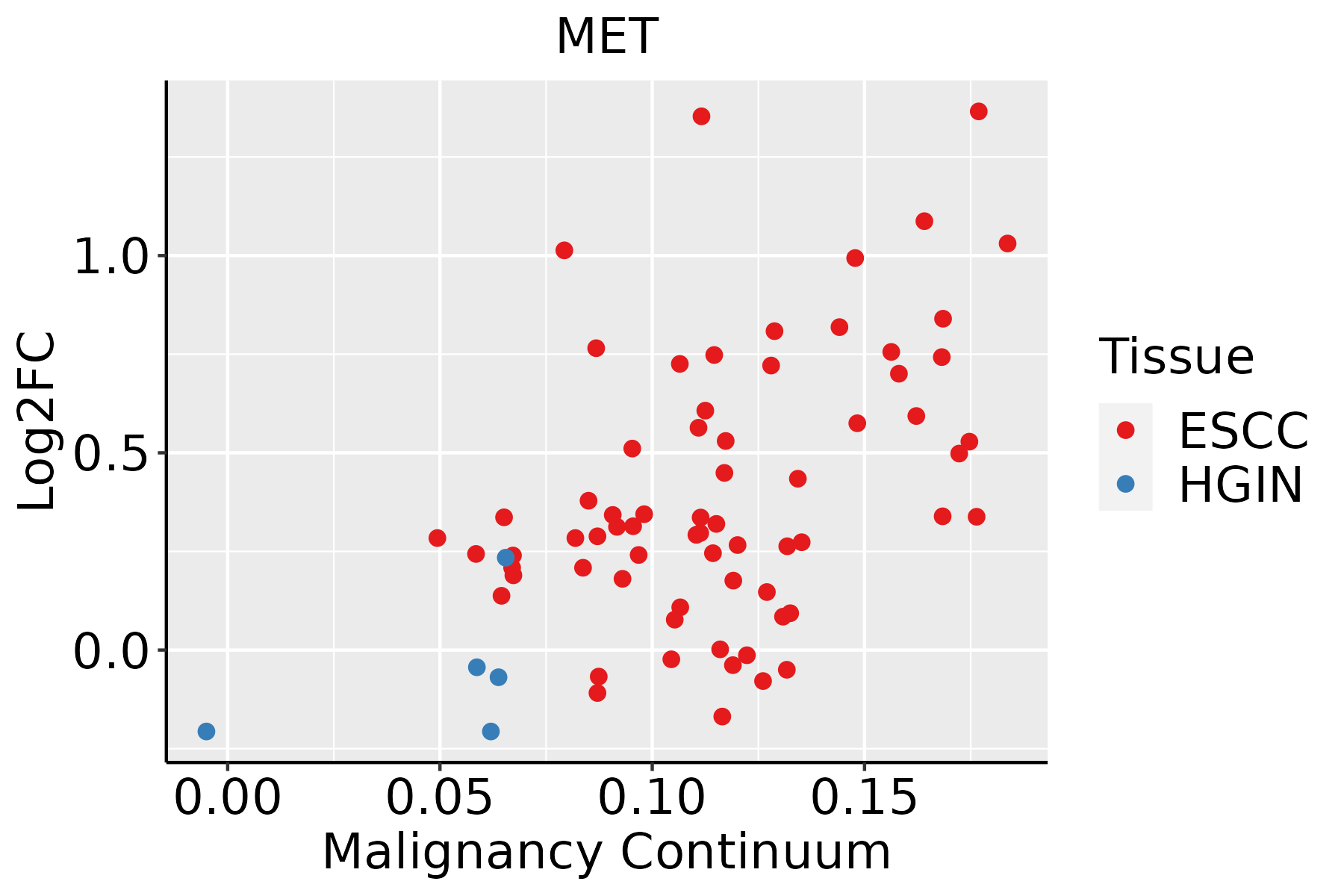 | ESCC: Esophageal squamous cell carcinoma |
| HGIN: High-grade intraepithelial neoplasias |
| LGIN: Low-grade intraepithelial neoplasias |
| Liver | 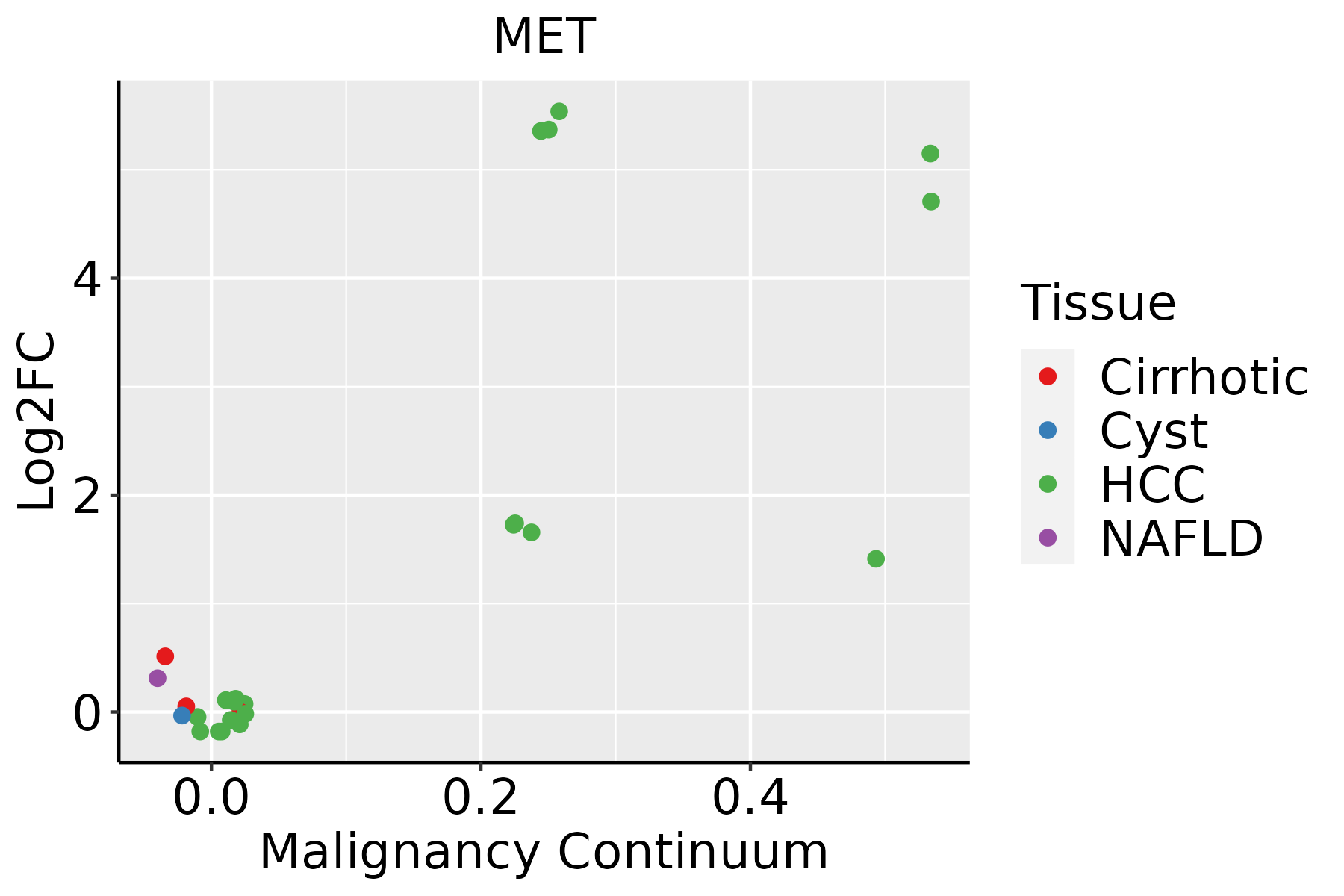 | HCC: Hepatocellular carcinoma |
| NAFLD: Non-alcoholic fatty liver disease |
| Lung | 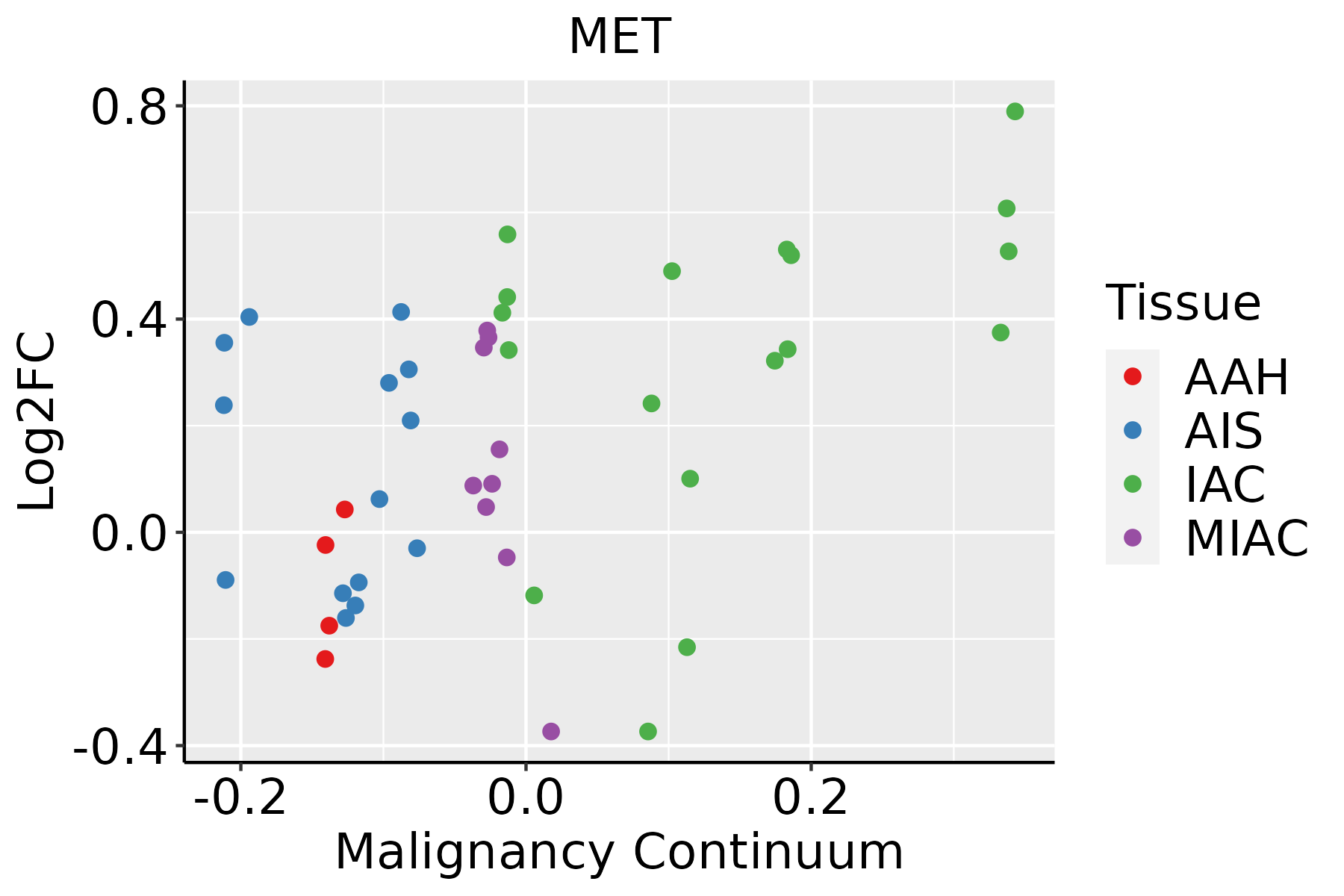 | AAH: Atypical adenomatous hyperplasia |
| AIS: Adenocarcinoma in situ |
| IAC: Invasive lung adenocarcinoma |
| MIA: Minimally invasive adenocarcinoma |
| Oral Cavity |  | EOLP: Erosive Oral lichen planus |
| LP: leukoplakia |
| NEOLP: Non-erosive oral lichen planus |
| OSCC: Oral squamous cell carcinoma |
| Prostate | 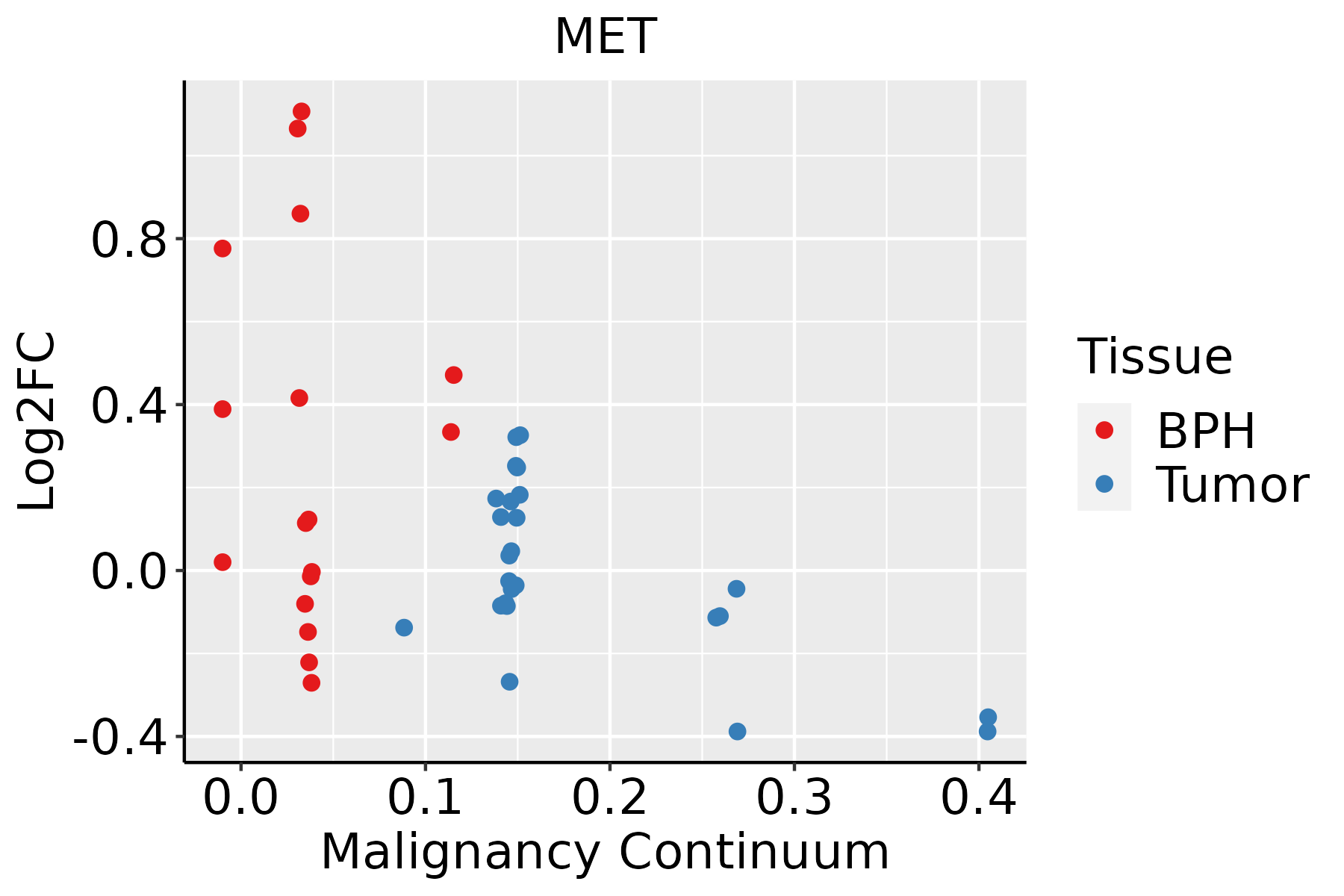 | BPH: Benign Prostatic Hyperplasia |
| Skin | 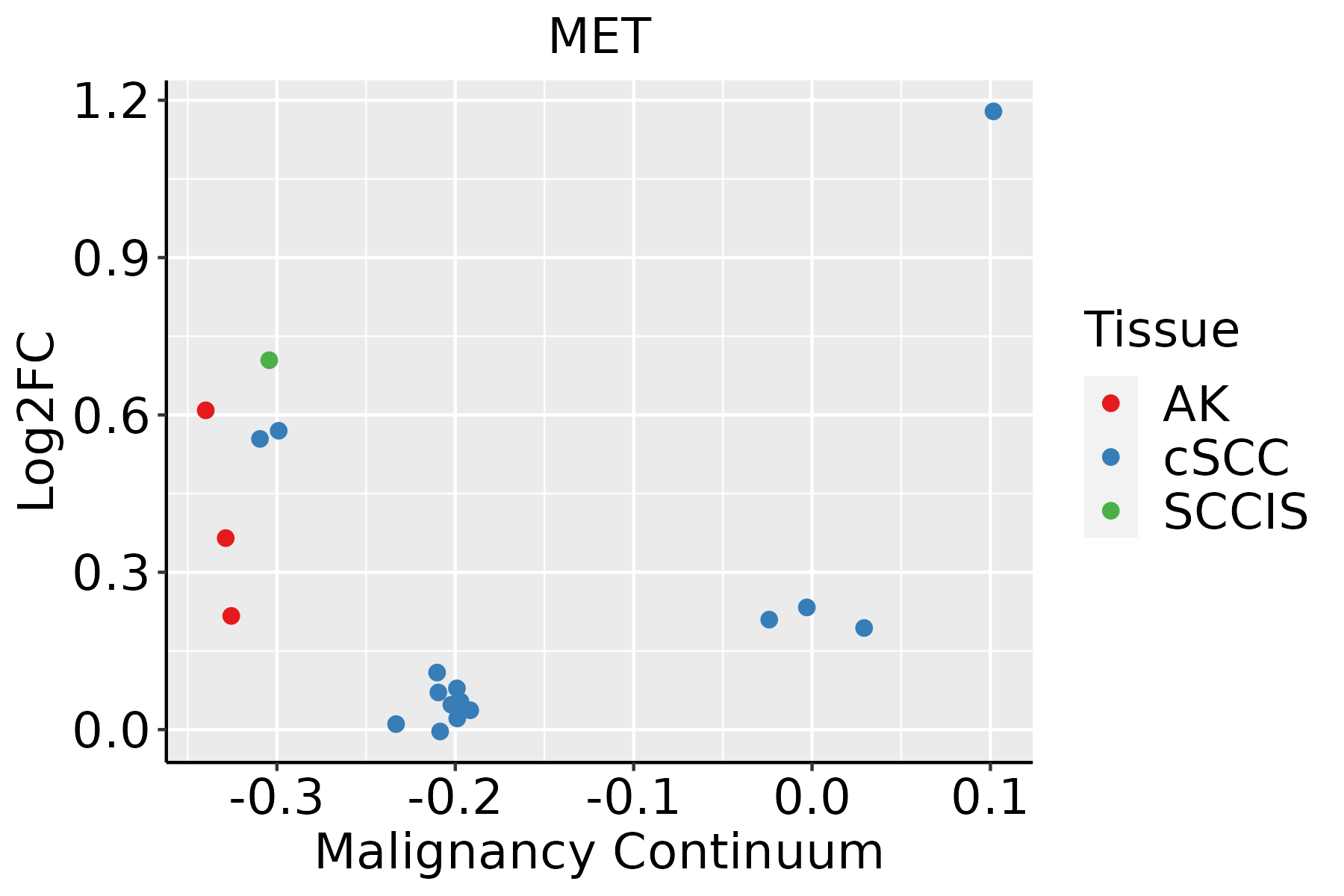 | AK: Actinic keratosis |
| cSCC: Cutaneous squamous cell carcinoma |
| SCCIS:squamous cell carcinoma in situ |
| Thyroid | 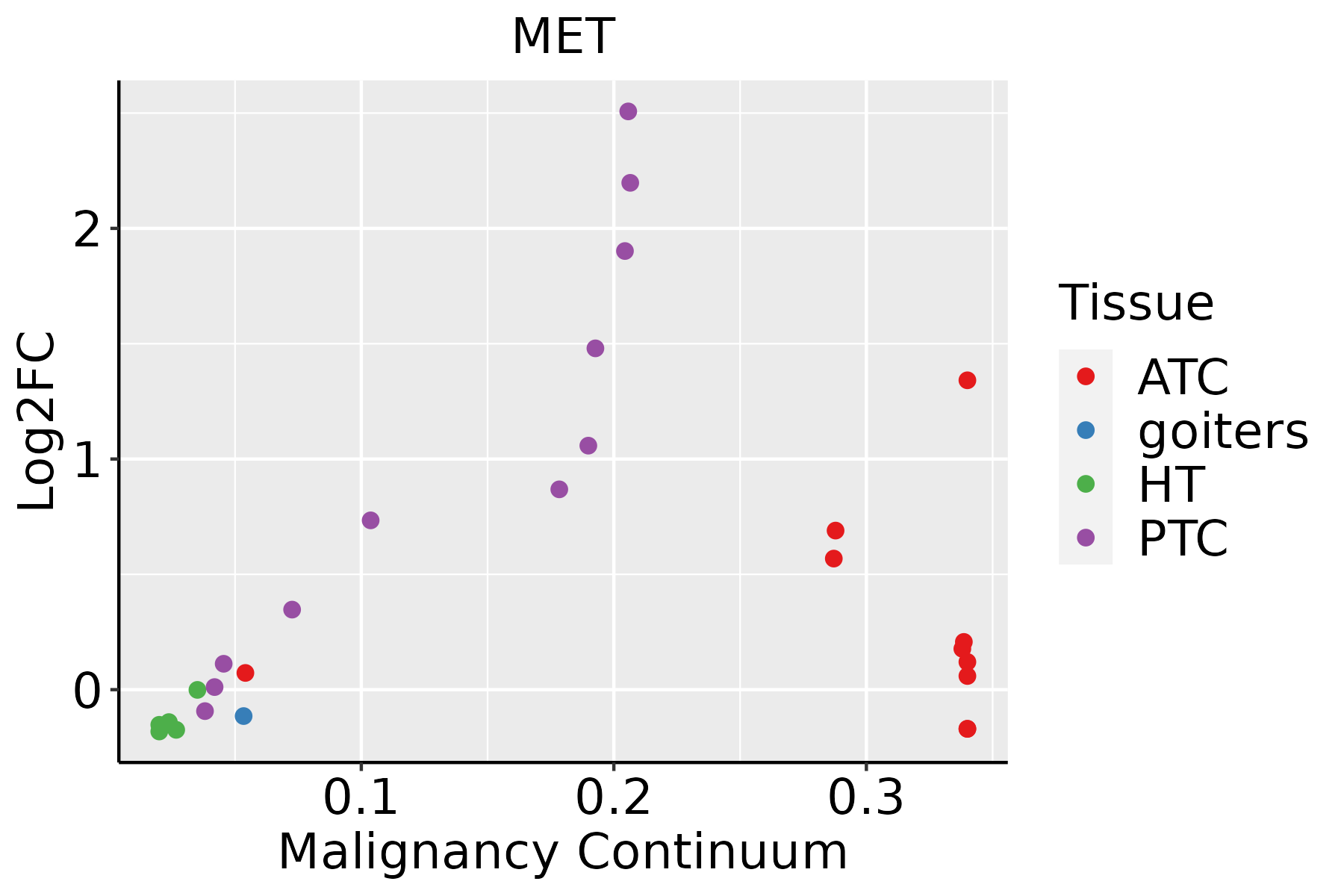 | ATC: Anaplastic thyroid cancer |
| HT: Hashimoto's thyroiditis |
| PTC: Papillary thyroid cancer |
| GO ID | Tissue | Disease Stage | Description | Gene Ratio | Bg Ratio | pvalue | p.adjust | Count |
| GO:004206311 | Breast | IDC | gliogenesis | 39/1434 | 301/18723 | 8.63e-04 | 1.00e-02 | 39 |
| GO:00100011 | Breast | IDC | glial cell differentiation | 31/1434 | 225/18723 | 1.04e-03 | 1.15e-02 | 31 |
| GO:00420632 | Breast | DCIS | gliogenesis | 39/1390 | 301/18723 | 4.76e-04 | 6.25e-03 | 39 |
| GO:00100012 | Breast | DCIS | glial cell differentiation | 31/1390 | 225/18723 | 6.24e-04 | 7.87e-03 | 31 |
| GO:0007015 | Colorectum | AD | actin filament organization | 167/3918 | 442/18723 | 1.65e-16 | 7.94e-14 | 167 |
| GO:0009895 | Colorectum | AD | negative regulation of catabolic process | 124/3918 | 320/18723 | 1.66e-13 | 3.35e-11 | 124 |
| GO:1902903 | Colorectum | AD | regulation of supramolecular fiber organization | 139/3918 | 383/18723 | 2.01e-12 | 3.19e-10 | 139 |
| GO:0031330 | Colorectum | AD | negative regulation of cellular catabolic process | 104/3918 | 262/18723 | 2.66e-12 | 3.96e-10 | 104 |
| GO:0032970 | Colorectum | AD | regulation of actin filament-based process | 142/3918 | 397/18723 | 4.05e-12 | 5.90e-10 | 142 |
| GO:0110053 | Colorectum | AD | regulation of actin filament organization | 108/3918 | 278/18723 | 5.02e-12 | 6.98e-10 | 108 |
| GO:0032271 | Colorectum | AD | regulation of protein polymerization | 94/3918 | 233/18723 | 1.01e-11 | 1.29e-09 | 94 |
| GO:0051258 | Colorectum | AD | protein polymerization | 112/3918 | 297/18723 | 1.85e-11 | 2.27e-09 | 112 |
| GO:0043254 | Colorectum | AD | regulation of protein-containing complex assembly | 148/3918 | 428/18723 | 2.64e-11 | 3.18e-09 | 148 |
| GO:0002064 | Colorectum | AD | epithelial cell development | 89/3918 | 220/18723 | 2.98e-11 | 3.52e-09 | 89 |
| GO:1902905 | Colorectum | AD | positive regulation of supramolecular fiber organization | 85/3918 | 209/18723 | 5.95e-11 | 6.33e-09 | 85 |
| GO:0048732 | Colorectum | AD | gland development | 149/3918 | 436/18723 | 6.07e-11 | 6.33e-09 | 149 |
| GO:0032956 | Colorectum | AD | regulation of actin cytoskeleton organization | 127/3918 | 358/18723 | 1.03e-10 | 1.04e-08 | 127 |
| GO:0051495 | Colorectum | AD | positive regulation of cytoskeleton organization | 89/3918 | 226/18723 | 1.61e-10 | 1.56e-08 | 89 |
| GO:0032273 | Colorectum | AD | positive regulation of protein polymerization | 62/3918 | 138/18723 | 1.94e-10 | 1.76e-08 | 62 |
| GO:0031334 | Colorectum | AD | positive regulation of protein-containing complex assembly | 91/3918 | 237/18723 | 4.78e-10 | 3.99e-08 | 91 |
| Hugo Symbol | Variant Class | Variant Classification | dbSNP RS | HGVSc | HGVSp | HGVSp Short | SWISSPROT | BIOTYPE | SIFT | PolyPhen | Tumor Sample Barcode | Tissue | Histology | Sex | Age | Stage | Therapy Types | Drugs | Outcome |
| MET | SNV | Missense_Mutation | rs752842662 | c.1406N>A | p.Arg469Gln | p.R469Q | P08581 | protein_coding | deleterious(0) | probably_damaging(1) | TCGA-AP-A0LM-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | <65 | III/IV | Chemotherapy | cisplatin | SD |
| MET | SNV | Missense_Mutation | novel | c.1604N>C | p.Phe535Ser | p.F535S | P08581 | protein_coding | deleterious(0) | probably_damaging(0.973) | TCGA-AP-A1DK-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | <65 | I/II | Unknown | Unknown | SD |
| MET | SNV | Missense_Mutation | novel | c.3065N>A | p.Arg1022Gln | p.R1022Q | P08581 | protein_coding | deleterious(0) | probably_damaging(1) | TCGA-AP-A1DM-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | <65 | I/II | Unknown | Unknown | SD |
| MET | SNV | Missense_Mutation | rs375951814 | c.1862N>T | p.Thr621Ile | p.T621I | P08581 | protein_coding | tolerated(0.21) | benign(0.028) | TCGA-AP-A1DV-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | <65 | I/II | Unknown | Unknown | SD |
| MET | SNV | Missense_Mutation | | c.1024N>A | p.Leu342Ile | p.L342I | P08581 | protein_coding | deleterious(0.01) | benign(0.356) | TCGA-AP-A1E0-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | <65 | III/IV | Chemotherapy | paclitaxel | SD |
| MET | SNV | Missense_Mutation | novel | c.1091N>T | p.Ala364Val | p.A364V | P08581 | protein_coding | tolerated(0.3) | possibly_damaging(0.481) | TCGA-AX-A06F-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | <65 | III/IV | Chemotherapy | carboplatin | SD |
| MET | SNV | Missense_Mutation | | c.1285T>G | p.Leu429Val | p.L429V | P08581 | protein_coding | deleterious(0.01) | probably_damaging(0.919) | TCGA-AX-A0J0-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | <65 | I/II | Unknown | Unknown | SD |
| MET | SNV | Missense_Mutation | rs367628460 | c.1234N>T | p.Arg412Cys | p.R412C | P08581 | protein_coding | tolerated(0.12) | possibly_damaging(0.707) | TCGA-AX-A0J1-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | >=65 | I/II | Unknown | Unknown | SD |
| MET | SNV | Missense_Mutation | | c.3143N>G | p.Asp1048Gly | p.D1048G | P08581 | protein_coding | tolerated(0.31) | probably_damaging(0.956) | TCGA-AX-A0J1-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | >=65 | I/II | Unknown | Unknown | SD |
| MET | SNV | Missense_Mutation | rs375951814 | c.1862N>T | p.Thr621Ile | p.T621I | P08581 | protein_coding | tolerated(0.21) | benign(0.028) | TCGA-AX-A1CE-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | <65 | I/II | Unspecific | Paclitaxel | SD |
| Entrez ID | Symbol | Category | Interaction Types | Drug Claim Name | Drug Name | PMIDs |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | | CBT-101 | | |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | inhibitor | 252166505 | TEPOTINIB | |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | inhibitor | 384403668 | | |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | | Ficlatuzumab | FICLATUZUMAB | 24901237 |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | inhibitor | CHEMBL2103851 | AMUVATINIB | |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | inhibitor | ARQ 197 | | |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | | ENTRECTINIB | ENTRECTINIB | |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | | Imatinib | IMATINIB | 28514312 |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | | TEPOTINIB | TEPOTINIB | 32469185,24061647 |
| 4233 | MET | KINASE, TYROSINE KINASE, DRUGGABLE GENOME, CLINICALLY ACTIONABLE, CELL SURFACE, DRUG RESISTANCE | | PALBOCICLIB | PALBOCICLIB | |







 Identification of the aberrant gene expression in precancerous and cancerous lesions by comparing the gene expression of stem-like cells in diseased tissues with normal stem cells
Identification of the aberrant gene expression in precancerous and cancerous lesions by comparing the gene expression of stem-like cells in diseased tissues with normal stem cells Find out the enriched GO biological processes and KEGG pathways involved in transition from healthy to precancer to cancer
Find out the enriched GO biological processes and KEGG pathways involved in transition from healthy to precancer to cancer
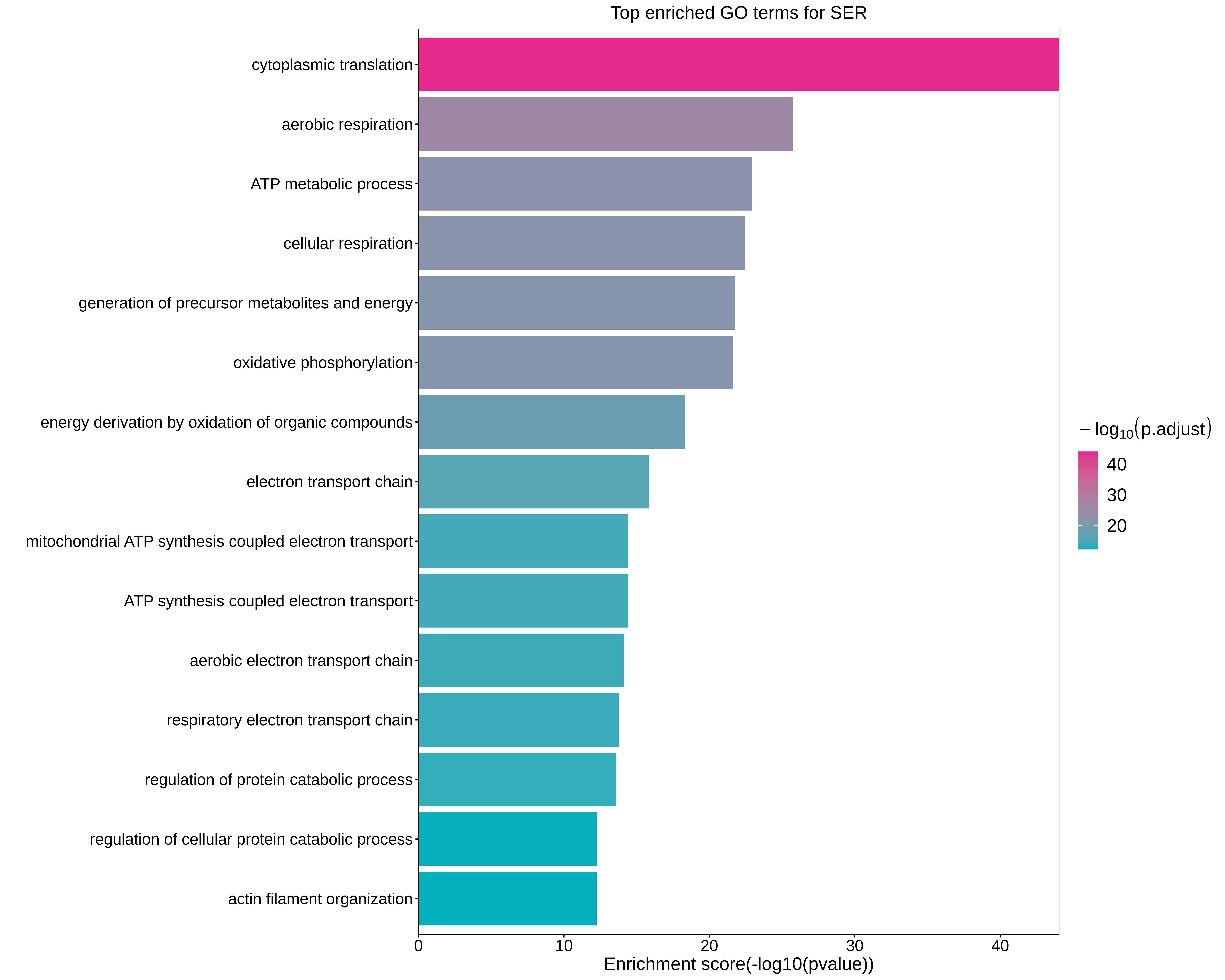

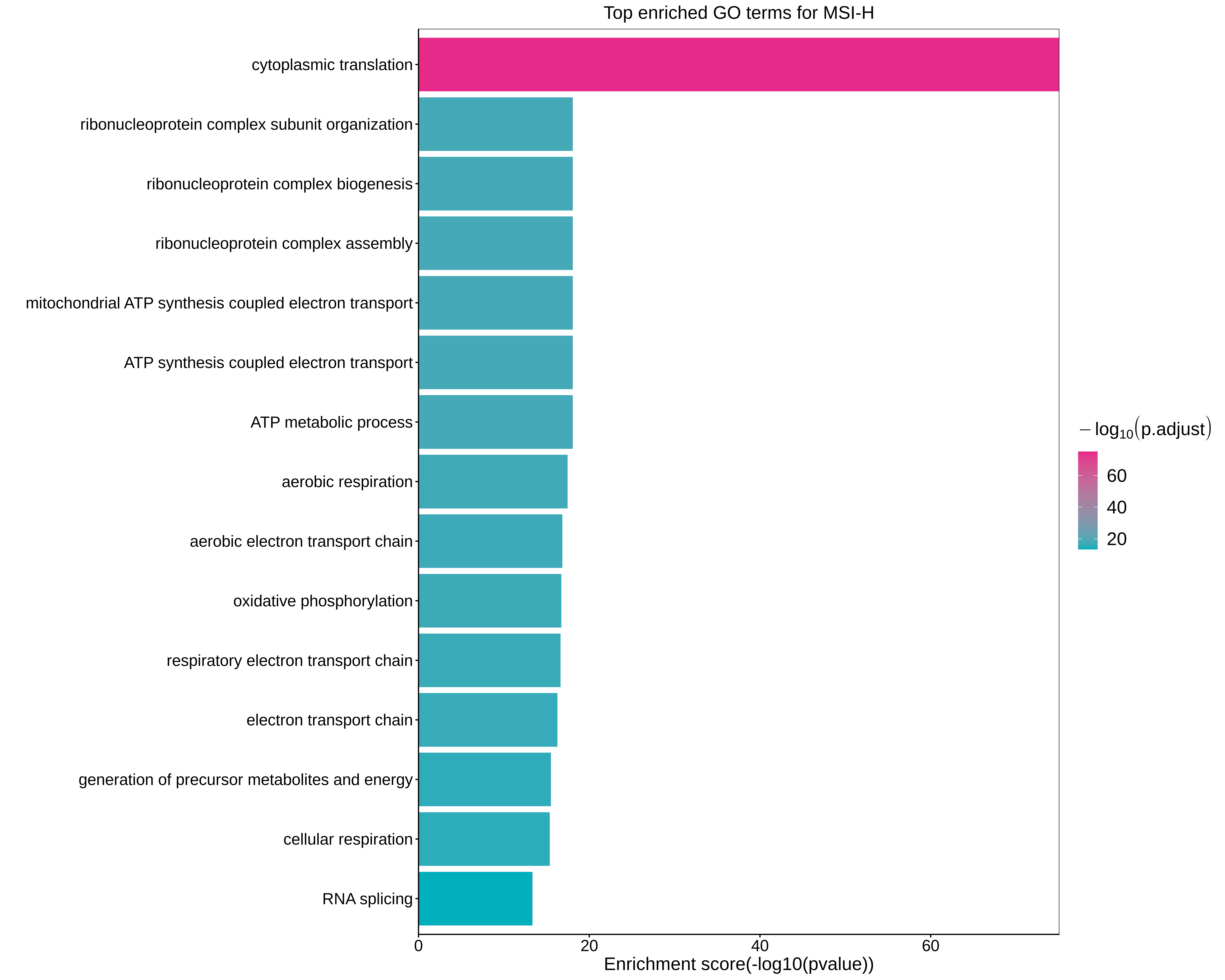
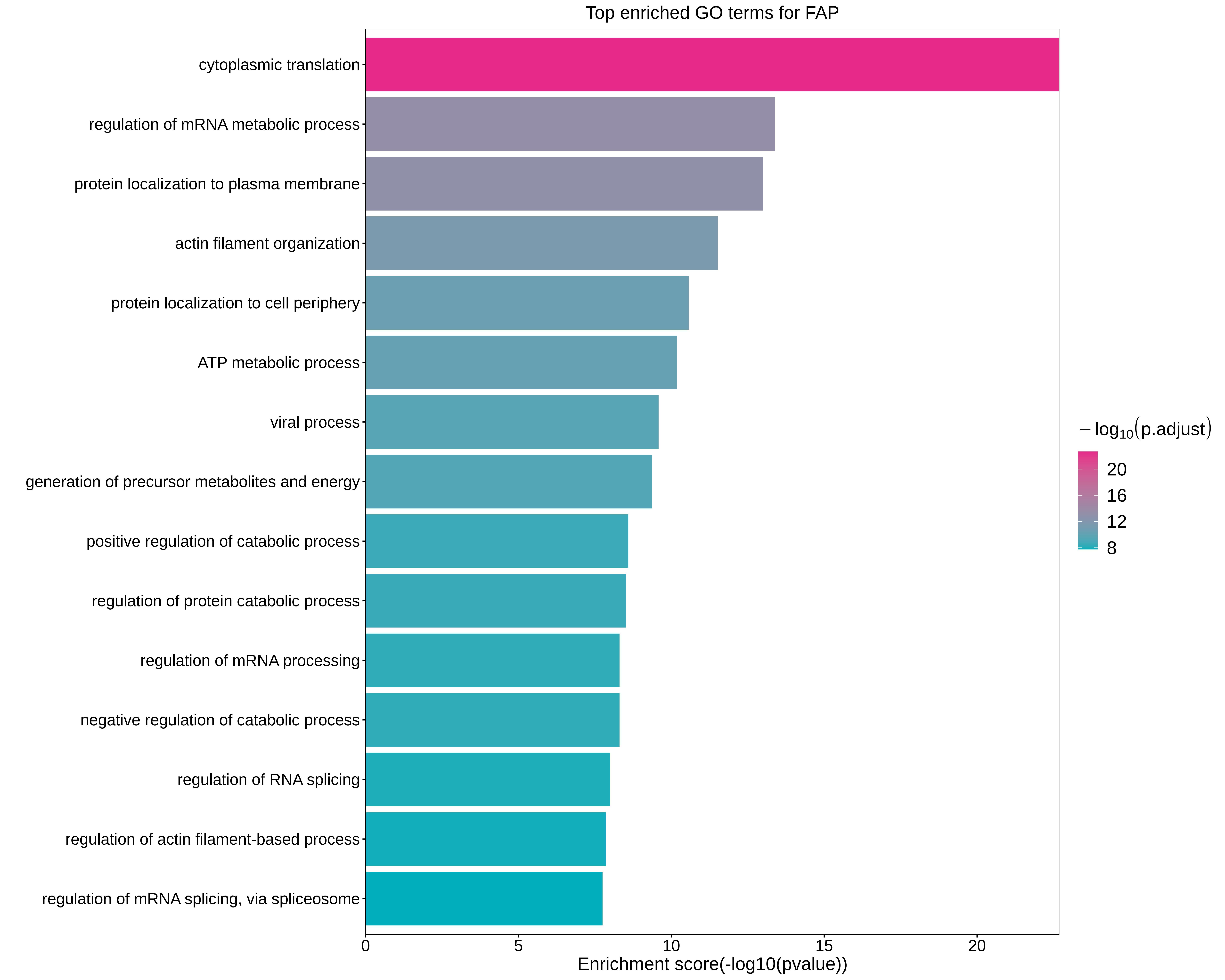
 Identification of potential cell-cell interactions between two cell types and their ligand-receptor pairs for different disease states
Identification of potential cell-cell interactions between two cell types and their ligand-receptor pairs for different disease states Find out the significant the regulons (TFs) and the target genes of each regulon across cell types for different disease states
Find out the significant the regulons (TFs) and the target genes of each regulon across cell types for different disease states Annotation of somatic variants for genes involved in malignant transformation
Annotation of somatic variants for genes involved in malignant transformation Identification of chemicals and drugs interact with genes involved in malignant transfromation
Identification of chemicals and drugs interact with genes involved in malignant transfromation








