| GO ID | Tissue | Disease Stage | Description | Gene Ratio | Bg Ratio | pvalue | p.adjust | Count |
| GO:00069139 | Cervix | CC | nucleocytoplasmic transport | 71/2311 | 301/18723 | 4.04e-08 | 2.54e-06 | 71 |
| GO:00511699 | Cervix | CC | nuclear transport | 71/2311 | 301/18723 | 4.04e-08 | 2.54e-06 | 71 |
| GO:000838010 | Cervix | CC | RNA splicing | 76/2311 | 434/18723 | 9.79e-04 | 8.19e-03 | 76 |
| GO:000037710 | Cervix | CC | RNA splicing, via transesterification reactions with bulged adenosine as nucleophile | 55/2311 | 320/18723 | 6.71e-03 | 3.70e-02 | 55 |
| GO:000039810 | Cervix | CC | mRNA splicing, via spliceosome | 55/2311 | 320/18723 | 6.71e-03 | 3.70e-02 | 55 |
| GO:000037510 | Cervix | CC | RNA splicing, via transesterification reactions | 55/2311 | 324/18723 | 8.61e-03 | 4.44e-02 | 55 |
| GO:0008380 | Colorectum | AD | RNA splicing | 169/3918 | 434/18723 | 3.59e-18 | 2.04e-15 | 169 |
| GO:0000377 | Colorectum | AD | RNA splicing, via transesterification reactions with bulged adenosine as nucleophile | 130/3918 | 320/18723 | 5.88e-16 | 2.16e-13 | 130 |
| GO:0000398 | Colorectum | AD | mRNA splicing, via spliceosome | 130/3918 | 320/18723 | 5.88e-16 | 2.16e-13 | 130 |
| GO:0000375 | Colorectum | AD | RNA splicing, via transesterification reactions | 131/3918 | 324/18723 | 7.11e-16 | 2.22e-13 | 131 |
| GO:0006913 | Colorectum | AD | nucleocytoplasmic transport | 102/3918 | 301/18723 | 1.00e-07 | 4.85e-06 | 102 |
| GO:0051169 | Colorectum | AD | nuclear transport | 102/3918 | 301/18723 | 1.00e-07 | 4.85e-06 | 102 |
| GO:0000380 | Colorectum | AD | alternative mRNA splicing, via spliceosome | 33/3918 | 77/18723 | 1.13e-05 | 2.55e-04 | 33 |
| GO:00083801 | Colorectum | SER | RNA splicing | 123/2897 | 434/18723 | 3.84e-12 | 8.41e-10 | 123 |
| GO:00003751 | Colorectum | SER | RNA splicing, via transesterification reactions | 90/2897 | 324/18723 | 8.68e-09 | 7.50e-07 | 90 |
| GO:00003771 | Colorectum | SER | RNA splicing, via transesterification reactions with bulged adenosine as nucleophile | 89/2897 | 320/18723 | 9.82e-09 | 8.14e-07 | 89 |
| GO:00003981 | Colorectum | SER | mRNA splicing, via spliceosome | 89/2897 | 320/18723 | 9.82e-09 | 8.14e-07 | 89 |
| GO:00069131 | Colorectum | SER | nucleocytoplasmic transport | 77/2897 | 301/18723 | 3.39e-06 | 1.32e-04 | 77 |
| GO:00511691 | Colorectum | SER | nuclear transport | 77/2897 | 301/18723 | 3.39e-06 | 1.32e-04 | 77 |
| GO:00003801 | Colorectum | SER | alternative mRNA splicing, via spliceosome | 24/2897 | 77/18723 | 4.08e-04 | 6.09e-03 | 24 |
| Pathway ID | Tissue | Disease Stage | Description | Gene Ratio | Bg Ratio | pvalue | p.adjust | qvalue | Count |
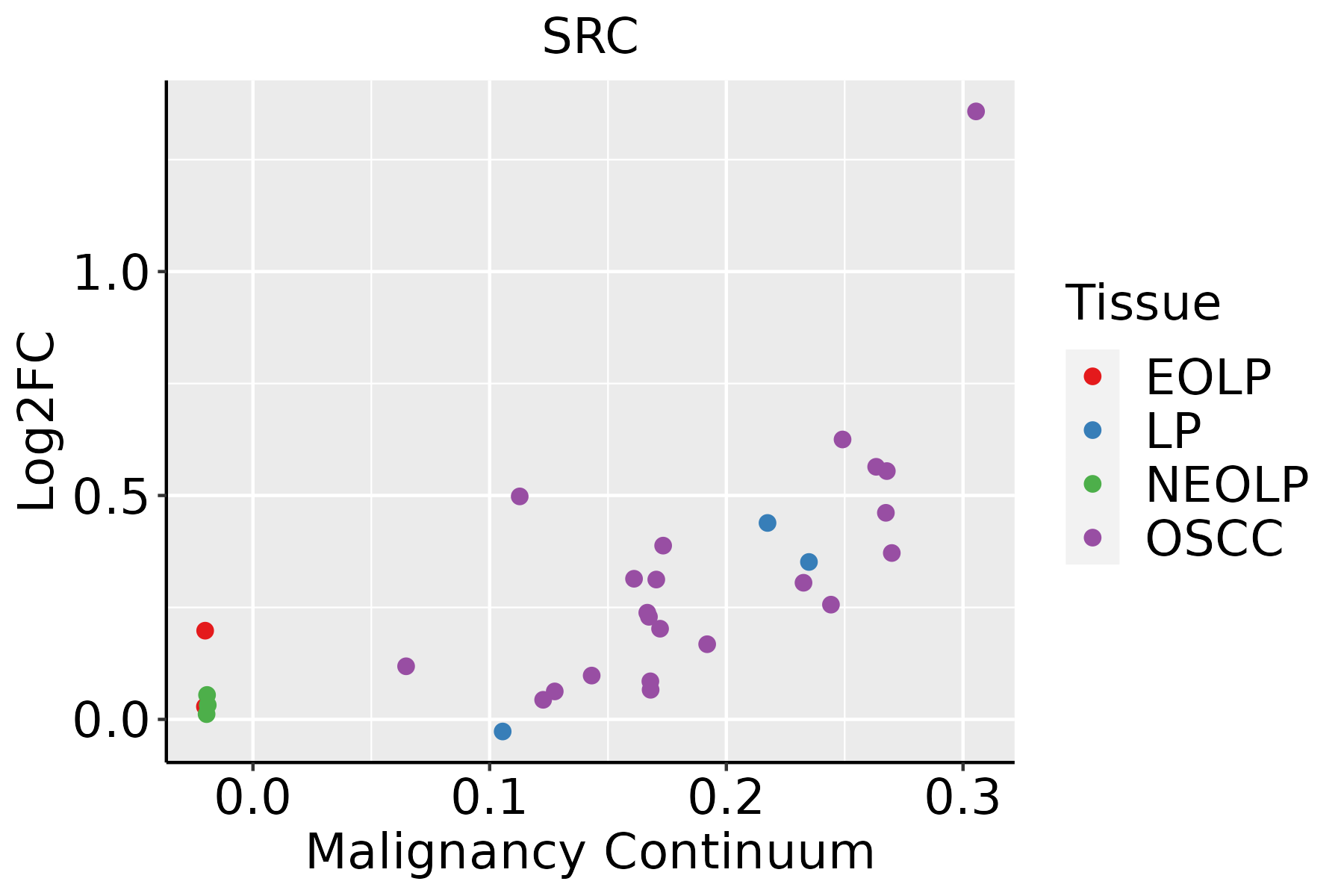
| hsa0513525 | Oral cavity | LP | Yersinia infection | 59/2418 | 137/8465 | 1.81e-04 | 1.04e-03 | 6.71e-04 | 59 |
| hsa05417210 | Oral cavity | LP | Lipid and atherosclerosis | 84/2418 | 215/8465 | 5.02e-04 | 2.61e-03 | 1.68e-03 | 84 |
| hsa0521923 | Oral cavity | LP | Bladder cancer | 22/2418 | 41/8465 | 6.22e-04 | 3.19e-03 | 2.06e-03 | 22 |
| hsa0516122 | Oral cavity | LP | Hepatitis B | 65/2418 | 162/8465 | 9.31e-04 | 4.43e-03 | 2.86e-03 | 65 |
| hsa0516329 | Oral cavity | LP | Human cytomegalovirus infection | 85/2418 | 225/8465 | 1.55e-03 | 6.89e-03 | 4.45e-03 | 85 |
| hsa0520527 | Oral cavity | LP | Proteoglycans in cancer | 77/2418 | 205/8465 | 3.01e-03 | 1.29e-02 | 8.35e-03 | 77 |
| hsa0462521 | Oral cavity | LP | C-type lectin receptor signaling pathway | 43/2418 | 104/8465 | 3.33e-03 | 1.41e-02 | 9.06e-03 | 43 |
| hsa0515221 | Oral cavity | LP | Tuberculosis | 65/2418 | 180/8465 | 1.60e-02 | 4.92e-02 | 3.17e-02 | 65 |
| hsa0520838 | Oral cavity | LP | Chemical carcinogenesis - reactive oxygen species | 119/2418 | 223/8465 | 3.00e-15 | 8.32e-14 | 5.37e-14 | 119 |
| hsa0414436 | Oral cavity | LP | Endocytosis | 121/2418 | 251/8465 | 1.89e-11 | 3.70e-10 | 2.38e-10 | 121 |
| hsa0520334 | Oral cavity | LP | Viral carcinogenesis | 90/2418 | 204/8465 | 1.20e-06 | 1.38e-05 | 8.89e-06 | 90 |
| hsa0513038 | Oral cavity | LP | Pathogenic Escherichia coli infection | 87/2418 | 197/8465 | 1.71e-06 | 1.78e-05 | 1.15e-05 | 87 |
| hsa0541838 | Oral cavity | LP | Fluid shear stress and atherosclerosis | 65/2418 | 139/8465 | 3.42e-06 | 3.26e-05 | 2.10e-05 | 65 |
| hsa0513138 | Oral cavity | LP | Shigellosis | 103/2418 | 247/8465 | 4.98e-06 | 4.14e-05 | 2.67e-05 | 103 |
| hsa0512034 | Oral cavity | LP | Epithelial cell signaling in Helicobacter pylori infection | 38/2418 | 70/8465 | 5.17e-06 | 4.20e-05 | 2.71e-05 | 38 |
| hsa0516737 | Oral cavity | LP | Kaposi sarcoma-associated herpesvirus infection | 84/2418 | 194/8465 | 6.66e-06 | 5.16e-05 | 3.33e-05 | 84 |
| hsa0413737 | Oral cavity | LP | Mitophagy - animal | 38/2418 | 72/8465 | 1.24e-05 | 8.63e-05 | 5.57e-05 | 38 |
| hsa0510038 | Oral cavity | LP | Bacterial invasion of epithelial cells | 39/2418 | 77/8465 | 3.34e-05 | 2.22e-04 | 1.43e-04 | 39 |
| hsa0513535 | Oral cavity | LP | Yersinia infection | 59/2418 | 137/8465 | 1.81e-04 | 1.04e-03 | 6.71e-04 | 59 |
| hsa0541738 | Oral cavity | LP | Lipid and atherosclerosis | 84/2418 | 215/8465 | 5.02e-04 | 2.61e-03 | 1.68e-03 | 84 |
| Hugo Symbol | Variant Class | Variant Classification | dbSNP RS | HGVSc | HGVSp | HGVSp Short | SWISSPROT | BIOTYPE | SIFT | PolyPhen | Tumor Sample Barcode | Tissue | Histology | Sex | Age | Stage | Therapy Types | Drugs | Outcome |
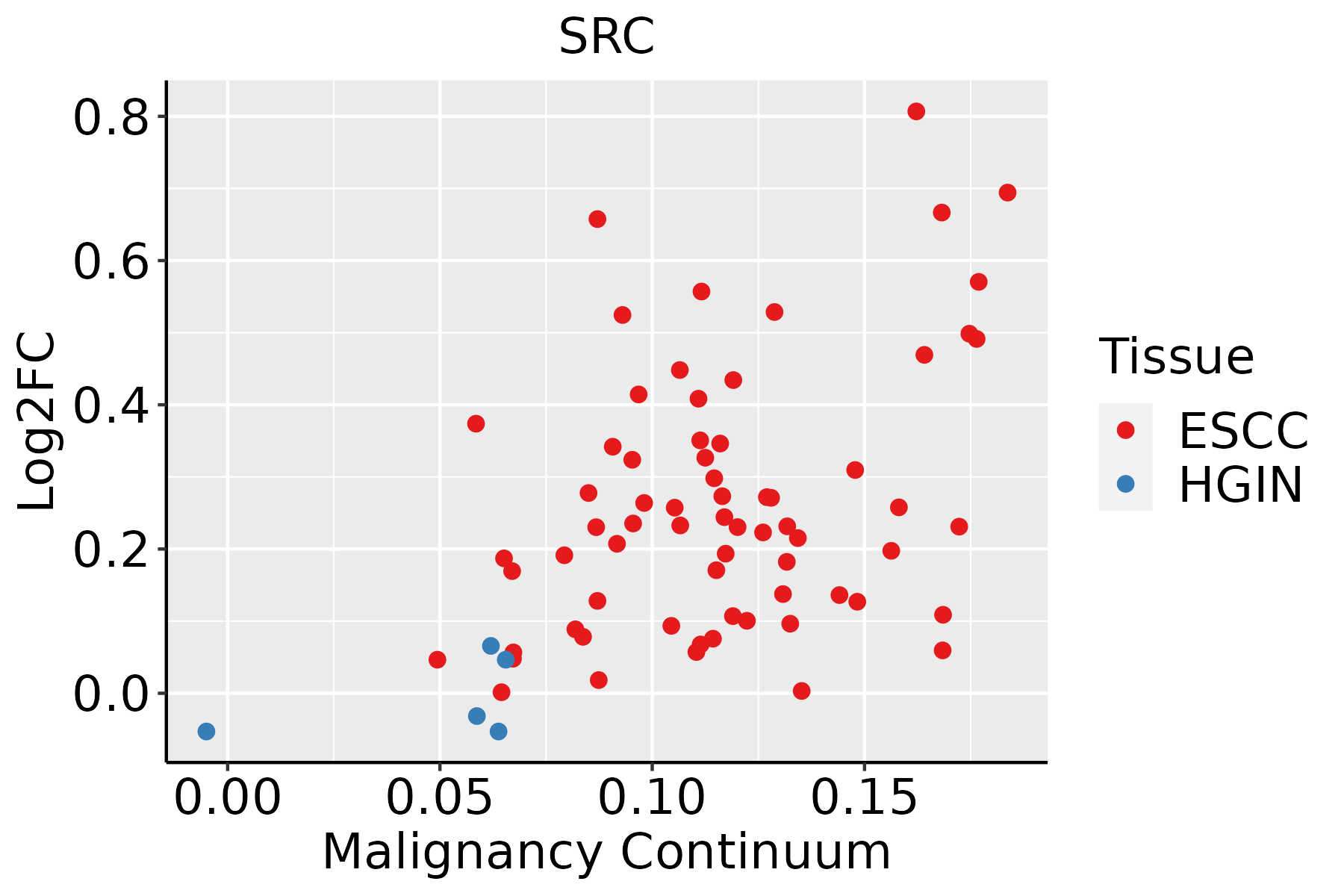
| SRC | SNV | Missense_Mutation | | c.806C>T | p.Ser269Leu | p.S269L | P12931 | protein_coding | deleterious(0) | probably_damaging(0.99) | TCGA-D8-A1J8-01 | Breast | breast invasive carcinoma | Female | >=65 | I/II | Hormone Therapy | nolvadex | SD |
| SRC | insertion | Frame_Shift_Ins | novel | c.1176_1177insCTCATCT | p.Ala393LeufsTer25 | p.A393Lfs*25 | P12931 | protein_coding | | | TCGA-AO-A0JB-01 | Breast | breast invasive carcinoma | Female | <65 | III/IV | Chemotherapy | cyclophosphamide | SD |
| SRC | insertion | Frame_Shift_Ins | novel | c.1177_1178insTGTCTGGGTCCGCTGGGGCCTCTTTC | p.Ala393ValfsTer72 | p.A393Vfs*72 | P12931 | protein_coding | | | TCGA-AO-A0JB-01 | Breast | breast invasive carcinoma | Female | <65 | III/IV | Chemotherapy | cyclophosphamide | SD |
| SRC | SNV | Missense_Mutation | novel | c.11N>G | p.Asn4Ser | p.N4S | P12931 | protein_coding | tolerated_low_confidence(0.97) | benign(0) | TCGA-2W-A8YY-01 | Cervix | cervical & endocervical cancer | Female | <65 | I/II | Chemotherapy | cisplatin | CR |
| SRC | SNV | Missense_Mutation | novel | c.439C>A | p.Gln147Lys | p.Q147K | P12931 | protein_coding | deleterious(0.04) | possibly_damaging(0.882) | TCGA-AA-3947-01 | Colorectum | colon adenocarcinoma | Female | <65 | I/II | Unknown | Unknown | SD |
| SRC | SNV | Missense_Mutation | | c.1571N>T | p.Thr524Met | p.T524M | P12931 | protein_coding | deleterious(0.01) | possibly_damaging(0.52) | TCGA-AD-6899-01 | Colorectum | colon adenocarcinoma | Male | >=65 | III/IV | Unknown | Unknown | SD |
| SRC | SNV | Missense_Mutation | | c.1489N>A | p.Leu497Ile | p.L497I | P12931 | protein_coding | tolerated(1) | probably_damaging(0.993) | TCGA-AD-A5EJ-01 | Colorectum | colon adenocarcinoma | Female | >=65 | I/II | Unknown | Unknown | SD |
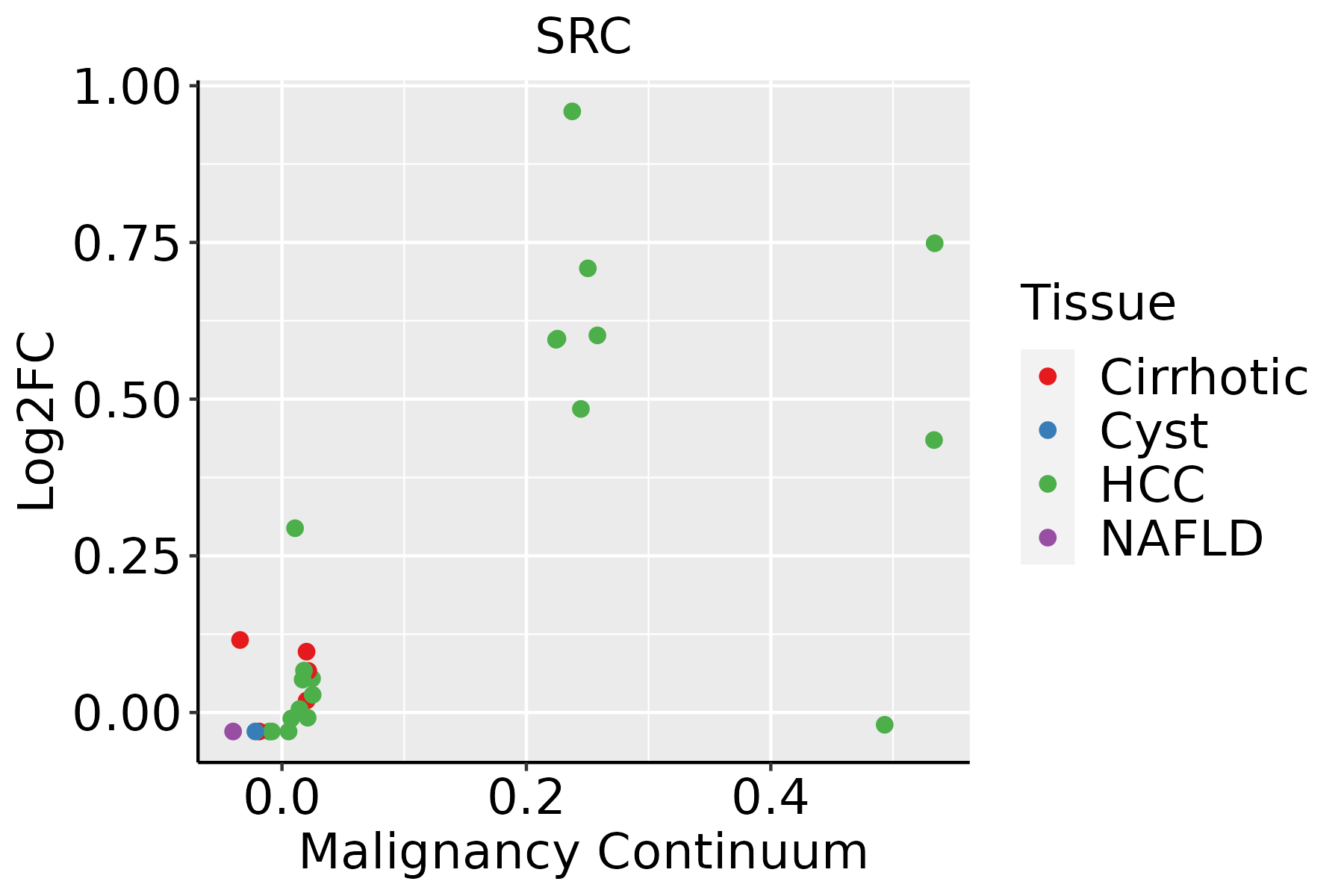
| SRC | SNV | Missense_Mutation | novel | c.659N>A | p.Arg220His | p.R220H | P12931 | protein_coding | deleterious(0.01) | benign(0.428) | TCGA-A5-A0G2-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | <65 | III/IV | Unknown | Unknown | SD |
| SRC | SNV | Missense_Mutation | rs533607141 | c.812N>A | p.Arg271Gln | p.R271Q | P12931 | protein_coding | tolerated(0.53) | benign(0.318) | TCGA-AJ-A3BG-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | >=65 | I/II | Chemotherapy | carboplatin | PD |
| SRC | SNV | Missense_Mutation | | c.272C>T | p.Ala91Val | p.A91V | P12931 | protein_coding | deleterious(0.01) | benign(0.437) | TCGA-AP-A059-01 | Endometrium | uterine corpus endometrioid carcinoma | Female | >=65 | I/II | Unknown | Unknown | SD |
| Entrez ID | Symbol | Category | Interaction Types | Drug Claim Name | Drug Name | PMIDs |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | | CEDIRANIB | CEDIRANIB | |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | inhibitor | CHEMBL24828 | VANDETANIB | |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | inhibitor | CHEMBL3545196 | PD-0166285 | |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | | DOVITINIB | DOVITINIB | |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | | TYRPHOSTIN A9 | | 11112699 |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | | KX2-361 | KX2-361 | |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | | AZD0530 | SARACATINIB | |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | inhibitor | 178102599 | | |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | inhibitor | CHEMBL1421 | DASATINIB | |
| 6714 | SRC | CLINICALLY ACTIONABLE, KINASE, TYROSINE KINASE, DRUGGABLE GENOME, TRANSCRIPTION FACTOR, ENZYME | inhibitor | 354702286 | | |







 Identification of the aberrant gene expression in precancerous and cancerous lesions by comparing the gene expression of stem-like cells in diseased tissues with normal stem cells
Identification of the aberrant gene expression in precancerous and cancerous lesions by comparing the gene expression of stem-like cells in diseased tissues with normal stem cells Find out the enriched GO biological processes and KEGG pathways involved in transition from healthy to precancer to cancer
Find out the enriched GO biological processes and KEGG pathways involved in transition from healthy to precancer to cancer
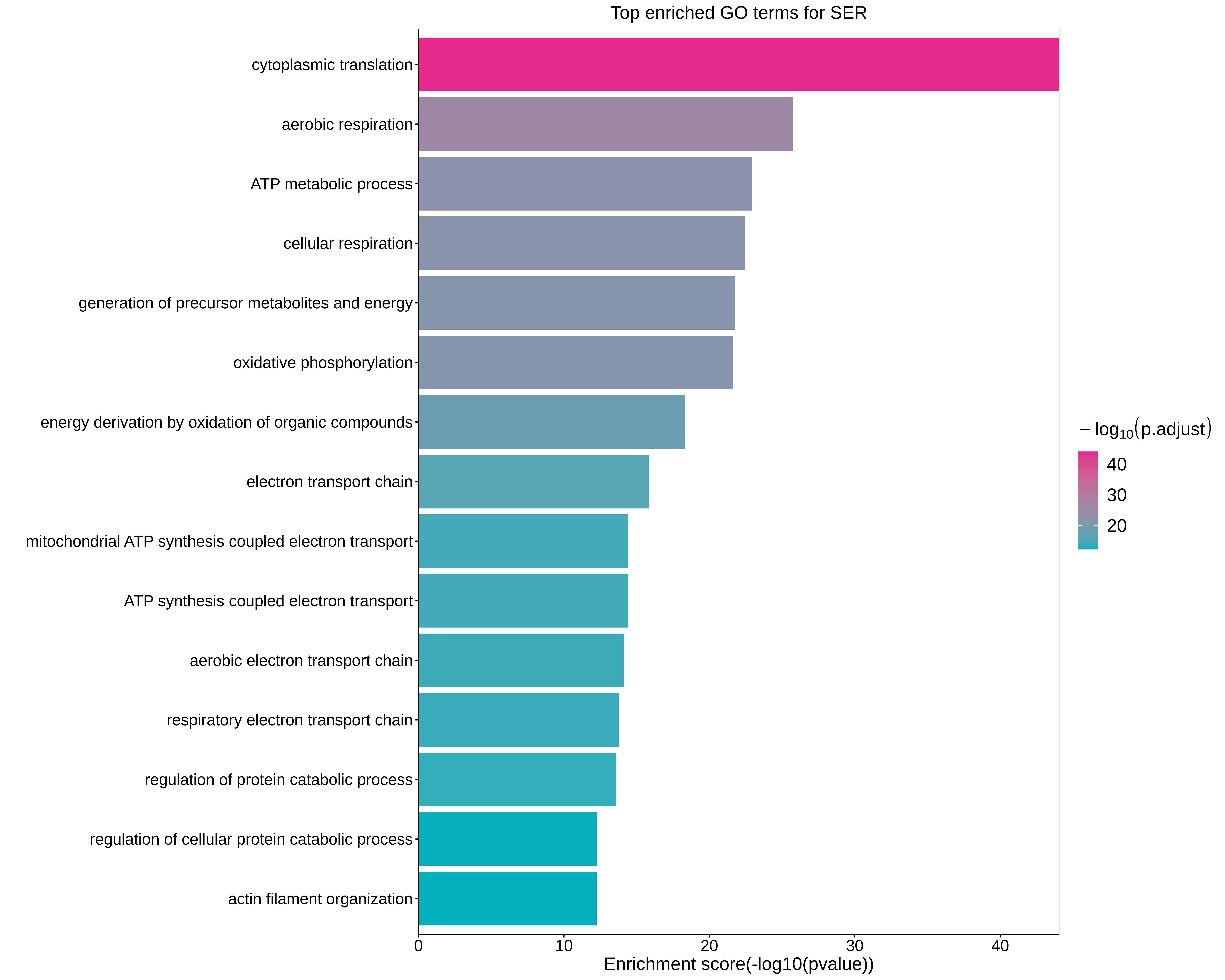

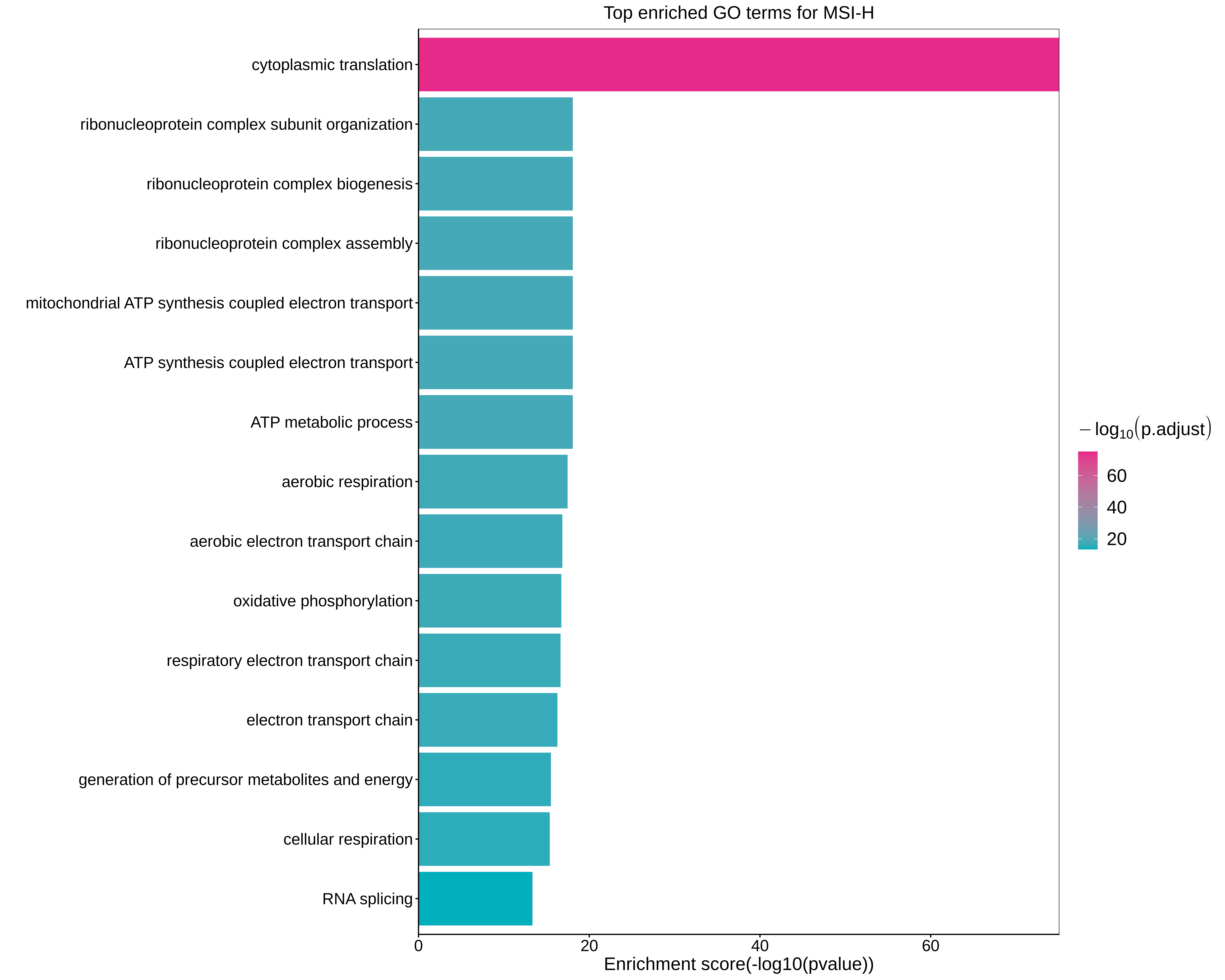
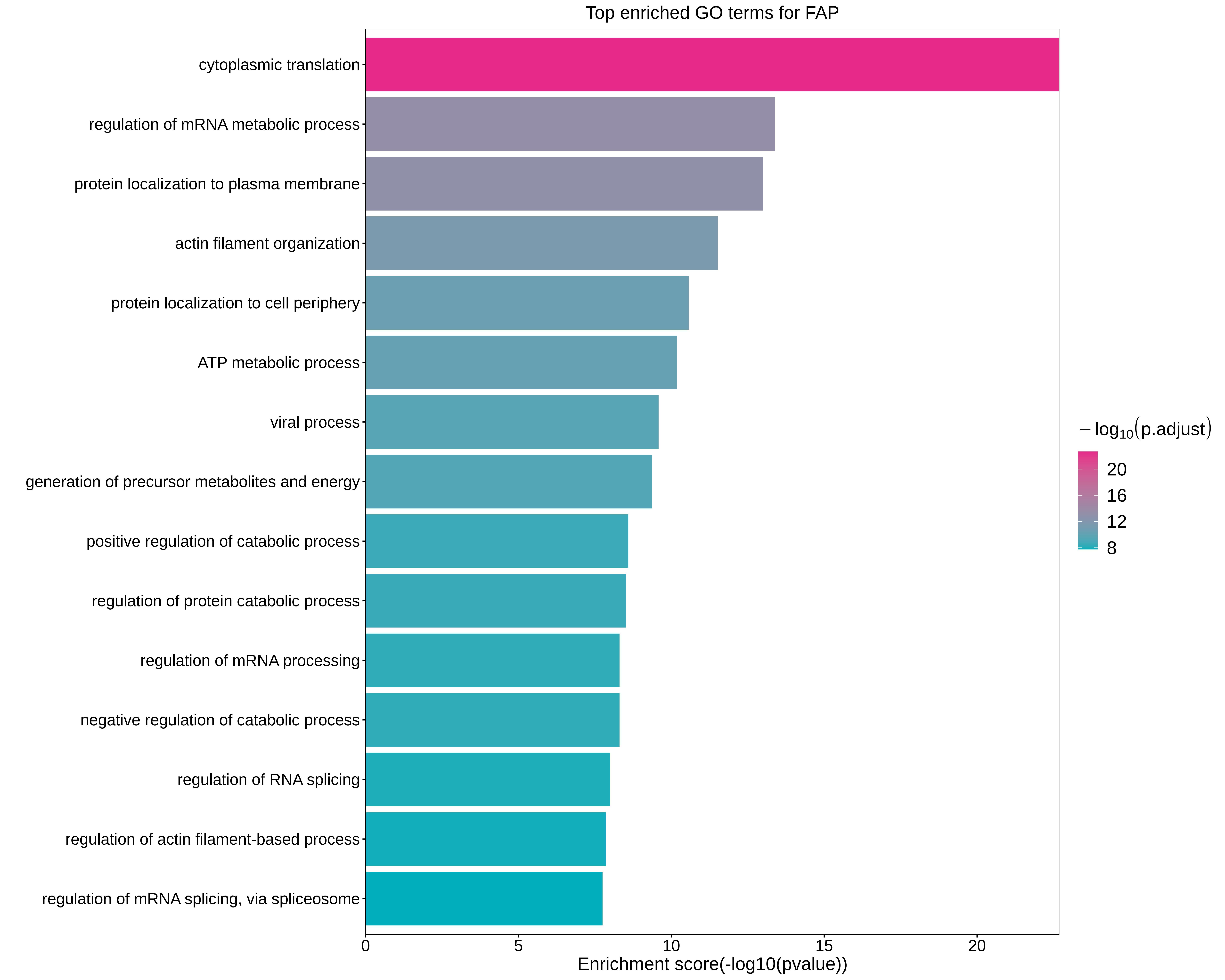
 Identification of potential cell-cell interactions between two cell types and their ligand-receptor pairs for different disease states
Identification of potential cell-cell interactions between two cell types and their ligand-receptor pairs for different disease states Find out the significant the regulons (TFs) and the target genes of each regulon across cell types for different disease states
Find out the significant the regulons (TFs) and the target genes of each regulon across cell types for different disease states Annotation of somatic variants for genes involved in malignant transformation
Annotation of somatic variants for genes involved in malignant transformation Identification of chemicals and drugs interact with genes involved in malignant transfromation
Identification of chemicals and drugs interact with genes involved in malignant transfromation