| Exon skip ID | chr | Exons involved in exon skipping | Skipped exon |
| exon_skip_112666 | chr19 | 3668404:3668440:3669673:3669904:3700297:3700390 | 3669673:3669904 |
| exon_skip_124949 | chr19 | 3647338:3647386:3648625:3648708:3651826:3652031 | 3648625:3648708 |
| exon_skip_176063 | chr19 | 3641705:3641809:3642907:3642939:3643243:3643381 | 3642907:3642939 |
| exon_skip_187868 | chr19 | 3661930:3662001:3664857:3664914:3667322:3667344 | 3664857:3664914 |
| exon_skip_206481 | chr19 | 3643243:3643381:3644087:3644251:3645974:3646058 | 3644087:3644251 |
| exon_skip_22139 | chr19 | 3632876:3633169:3633437:3633520:3638884:3639016 | 3633437:3633520 |
| exon_skip_294421 | chr19 | 3661930:3662001:3664822:3664914:3667322:3667344 | 3664822:3664914 |
| exon_skip_42845 | chr19 | 3633113:3633169:3633437:3633520:3638884:3639016 | 3633437:3633520 |
| exon_skip_6536 | chr19 | 3643249:3643381:3644087:3644251:3645974:3646058 | 3644087:3644251 |
| exon_skip_72313 | chr19 | 3644089:3644251:3645974:3646058:3647338:3647386 | 3645974:3646058 |
| UniProt acc. | Start of exon skipping (AA) | End of exon skipping (AA) | Protein feature start (AA) | Protein feature end (AA) | Category of protein feature | Description of feature |
| O60331 | 42 | 72 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 376 | 403 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 376 | 403 | 75 | 443 | Domain | Note=PIPK;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00781 |
| O60331 | 448 | 503 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 448 | 503 | 459 | 459 | Modified residue | Note=Asymmetric dimethylarginine%3B alternate;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:O70161 |
| O60331 | 448 | 503 | 459 | 459 | Modified residue | Note=Omega-N-methylarginine%3B alternate;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:O70161 |
| O60331 | 550 | 560 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 550 | 560 | 555 | 555 | Modified residue | Note=Phosphoserine;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:Q5I6B8 |
| O60331 | 640 | 667 | 640 | 668 | Alternative sequence | ID=VSP_042078;Note=In isoform 2. FPTDERSWVYSPLHYSAQAPPASDGESDT->FWRLWGPHAPTWPWRREGRAACLCPYPPHVVTPFPGTGLCASWSPDGTGGLGAMSCCVSVS;Ontology_term=ECO:0000303;evidence=ECO:0000303|PubMed:19548880;Dbxref=PMID:19548880 |
| O60331 | 640 | 667 | 641 | 668 | Alternative sequence | ID=VSP_042080;Note=In isoform 3. PTDERSWVYSPLHYSAQAPPASDGESDT->FTDGRYWIYSPRHRRLRAVTLSASGTVSDRSRPPWGEGAVPLGQQGAAGPRPEAQCLTSVVFQKGFG;Ontology_term=ECO:0000303;evidence=ECO:0000303|PubMed:19548880;Dbxref=PMID:19548880 |
| O60331 | 640 | 667 | 641 | 668 | Alternative sequence | ID=VSP_042079;Note=In isoform 4. Missing;Ontology_term=ECO:0000303;evidence=ECO:0000303|PubMed:14702039;Dbxref=PMID:14702039 |
| O60331 | 640 | 667 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 640 | 667 | 644 | 646 | Helix | Ontology_term=ECO:0000244;evidence=ECO:0000244|PDB:3H1Z |
| O60331 | 640 | 667 | 649 | 649 | Modified residue | Note=Phosphotyrosine%3B by CSK;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 650 | 650 | Modified residue | Note=Phosphoserine%3B by CDK5%2C MAPK1 and CDK1;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 662 | 662 | Modified residue | Note=Phosphoserine;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:O70161 |
| O60331 | 640 | 667 | 666 | 666 | Modified residue | Note=Phosphoserine;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:Q5I6B8 |
| O60331 | 640 | 667 | 650 | 650 | Mutagenesis | Note=Abolishes binding to TLN2. Affects localization to focal adhesions. S->D;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 650 | 650 | Mutagenesis | Note=Does not affect binding to TLN2 and localization to focal adhesions. S->N;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 641 | 668 | Region | Note=Mediates interaction with TLN2;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:12422219;Dbxref=PMID:12422219 |
| UniProt acc. | Start of exon skipping (AA) | End of exon skipping (AA) | Protein feature start (AA) | Protein feature end (AA) | Category of protein feature | Description of feature |
| O60331 | 42 | 72 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 376 | 403 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 376 | 403 | 75 | 443 | Domain | Note=PIPK;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00781 |
| O60331 | 550 | 560 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 550 | 560 | 555 | 555 | Modified residue | Note=Phosphoserine;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:Q5I6B8 |
| O60331 | 640 | 667 | 640 | 668 | Alternative sequence | ID=VSP_042078;Note=In isoform 2. FPTDERSWVYSPLHYSAQAPPASDGESDT->FWRLWGPHAPTWPWRREGRAACLCPYPPHVVTPFPGTGLCASWSPDGTGGLGAMSCCVSVS;Ontology_term=ECO:0000303;evidence=ECO:0000303|PubMed:19548880;Dbxref=PMID:19548880 |
| O60331 | 640 | 667 | 641 | 668 | Alternative sequence | ID=VSP_042080;Note=In isoform 3. PTDERSWVYSPLHYSAQAPPASDGESDT->FTDGRYWIYSPRHRRLRAVTLSASGTVSDRSRPPWGEGAVPLGQQGAAGPRPEAQCLTSVVFQKGFG;Ontology_term=ECO:0000303;evidence=ECO:0000303|PubMed:19548880;Dbxref=PMID:19548880 |
| O60331 | 640 | 667 | 641 | 668 | Alternative sequence | ID=VSP_042079;Note=In isoform 4. Missing;Ontology_term=ECO:0000303;evidence=ECO:0000303|PubMed:14702039;Dbxref=PMID:14702039 |
| O60331 | 640 | 667 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 640 | 667 | 644 | 646 | Helix | Ontology_term=ECO:0000244;evidence=ECO:0000244|PDB:3H1Z |
| O60331 | 640 | 667 | 649 | 649 | Modified residue | Note=Phosphotyrosine%3B by CSK;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 650 | 650 | Modified residue | Note=Phosphoserine%3B by CDK5%2C MAPK1 and CDK1;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 662 | 662 | Modified residue | Note=Phosphoserine;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:O70161 |
| O60331 | 640 | 667 | 666 | 666 | Modified residue | Note=Phosphoserine;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:Q5I6B8 |
| O60331 | 640 | 667 | 650 | 650 | Mutagenesis | Note=Abolishes binding to TLN2. Affects localization to focal adhesions. S->D;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 650 | 650 | Mutagenesis | Note=Does not affect binding to TLN2 and localization to focal adhesions. S->N;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 641 | 668 | Region | Note=Mediates interaction with TLN2;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:12422219;Dbxref=PMID:12422219 |
| UniProt acc. | Start of exon skipping (AA) | End of exon skipping (AA) | Protein feature start (AA) | Protein feature end (AA) | Category of protein feature | Description of feature |
| O60331 | 42 | 72 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 376 | 403 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 376 | 403 | 75 | 443 | Domain | Note=PIPK;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00781 |
| O60331 | 448 | 503 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 448 | 503 | 459 | 459 | Modified residue | Note=Asymmetric dimethylarginine%3B alternate;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:O70161 |
| O60331 | 448 | 503 | 459 | 459 | Modified residue | Note=Omega-N-methylarginine%3B alternate;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:O70161 |
| O60331 | 550 | 560 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 550 | 560 | 555 | 555 | Modified residue | Note=Phosphoserine;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:Q5I6B8 |
| O60331 | 640 | 667 | 640 | 668 | Alternative sequence | ID=VSP_042078;Note=In isoform 2. FPTDERSWVYSPLHYSAQAPPASDGESDT->FWRLWGPHAPTWPWRREGRAACLCPYPPHVVTPFPGTGLCASWSPDGTGGLGAMSCCVSVS;Ontology_term=ECO:0000303;evidence=ECO:0000303|PubMed:19548880;Dbxref=PMID:19548880 |
| O60331 | 640 | 667 | 641 | 668 | Alternative sequence | ID=VSP_042080;Note=In isoform 3. PTDERSWVYSPLHYSAQAPPASDGESDT->FTDGRYWIYSPRHRRLRAVTLSASGTVSDRSRPPWGEGAVPLGQQGAAGPRPEAQCLTSVVFQKGFG;Ontology_term=ECO:0000303;evidence=ECO:0000303|PubMed:19548880;Dbxref=PMID:19548880 |
| O60331 | 640 | 667 | 641 | 668 | Alternative sequence | ID=VSP_042079;Note=In isoform 4. Missing;Ontology_term=ECO:0000303;evidence=ECO:0000303|PubMed:14702039;Dbxref=PMID:14702039 |
| O60331 | 640 | 667 | 1 | 668 | Chain | ID=PRO_0000185462;Note=Phosphatidylinositol 4-phosphate 5-kinase type-1 gamma |
| O60331 | 640 | 667 | 644 | 646 | Helix | Ontology_term=ECO:0000244;evidence=ECO:0000244|PDB:3H1Z |
| O60331 | 640 | 667 | 649 | 649 | Modified residue | Note=Phosphotyrosine%3B by CSK;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 650 | 650 | Modified residue | Note=Phosphoserine%3B by CDK5%2C MAPK1 and CDK1;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 662 | 662 | Modified residue | Note=Phosphoserine;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:O70161 |
| O60331 | 640 | 667 | 666 | 666 | Modified residue | Note=Phosphoserine;Ontology_term=ECO:0000250;evidence=ECO:0000250|UniProtKB:Q5I6B8 |
| O60331 | 640 | 667 | 650 | 650 | Mutagenesis | Note=Abolishes binding to TLN2. Affects localization to focal adhesions. S->D;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 650 | 650 | Mutagenesis | Note=Does not affect binding to TLN2 and localization to focal adhesions. S->N;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:15738269;Dbxref=PMID:15738269 |
| O60331 | 640 | 667 | 641 | 668 | Region | Note=Mediates interaction with TLN2;Ontology_term=ECO:0000269;evidence=ECO:0000269|PubMed:12422219;Dbxref=PMID:12422219 |












 Gene summary
Gene summary Gene ontology of each this gene with evidence of Inferred from Direct Assay (IDA) from Entrez
Gene ontology of each this gene with evidence of Inferred from Direct Assay (IDA) from Entrez  Skipped exons in the ROSMAP, MSBB, and Mayo based on Ensembl gene isoform structure.
Skipped exons in the ROSMAP, MSBB, and Mayo based on Ensembl gene isoform structure. 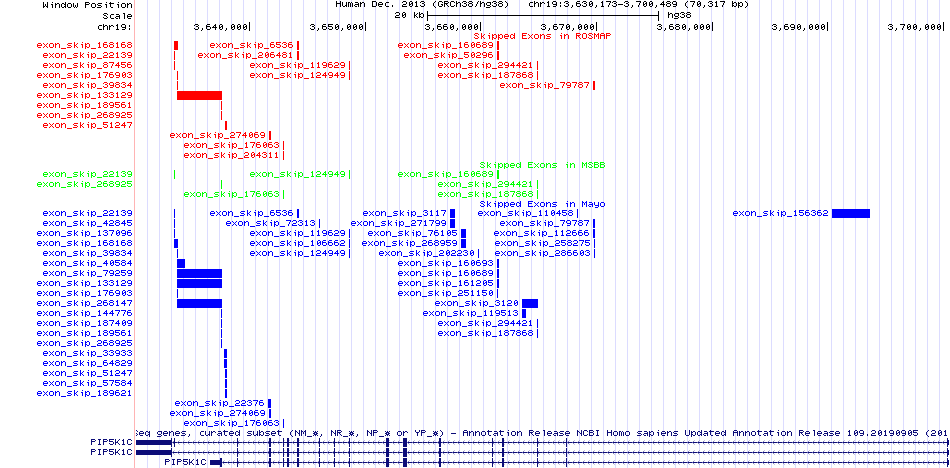
 Differentially expressed gene analysis across multiple brain tissues between AD and control.
Differentially expressed gene analysis across multiple brain tissues between AD and control. Differentially expressed isoform analysis across multiple brain tissues between AD and control.
Differentially expressed isoform analysis across multiple brain tissues between AD and control. Landscape of isoform expressions across multiple brain tissues between AD and control.
Landscape of isoform expressions across multiple brain tissues between AD and control. Landscape of individual exon skipping event across AD tissues and controls (PSI heatmap).
Landscape of individual exon skipping event across AD tissues and controls (PSI heatmap). All exon skipping events in AD cohorts.
All exon skipping events in AD cohorts. Differentially expressed PSI values of individual exon skipping events in multiple brain tissues between AD and control.
Differentially expressed PSI values of individual exon skipping events in multiple brain tissues between AD and control. Open reading frame (ORF) of individual exon skipping events in ROSMAP based on the Ensembl gene structure combined from isoforms.
Open reading frame (ORF) of individual exon skipping events in ROSMAP based on the Ensembl gene structure combined from isoforms. Open reading frame (ORF) of individual exon skipping events in MSBB based on the Ensembl gene structure combined from isoforms.
Open reading frame (ORF) of individual exon skipping events in MSBB based on the Ensembl gene structure combined from isoforms. Open reading frame (ORF) of individual exon skipping events in Mayo based on the Ensembl gene structure combined from isoforms.
Open reading frame (ORF) of individual exon skipping events in Mayo based on the Ensembl gene structure combined from isoforms.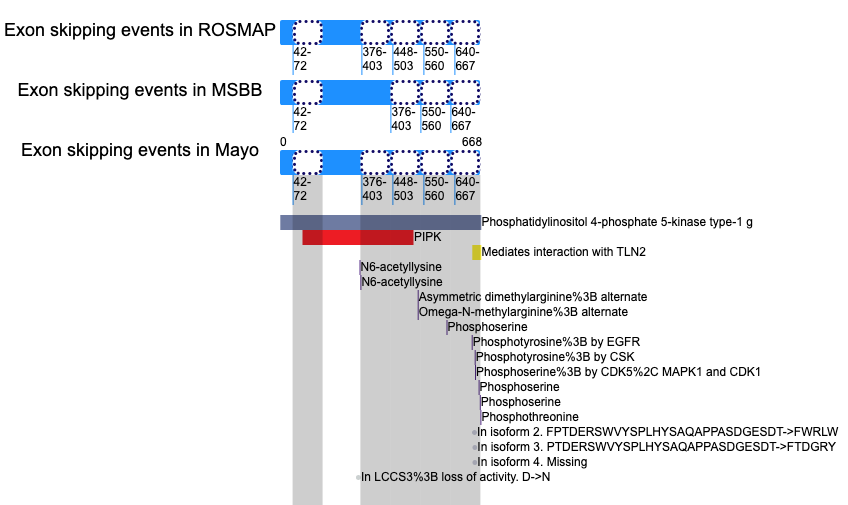
 Loci of skipped exons in ROSMAP across genomic, transcript, and protein sequence levels of In-frame cases.
Loci of skipped exons in ROSMAP across genomic, transcript, and protein sequence levels of In-frame cases. Loci of skipped exons in MSBB across genomic, transcript, and protein sequence levels of In-frame cases.
Loci of skipped exons in MSBB across genomic, transcript, and protein sequence levels of In-frame cases. Loci of skipped exons in Mayo across genomic, transcript, and protein sequence levels of In-frame cases.
Loci of skipped exons in Mayo across genomic, transcript, and protein sequence levels of In-frame cases. Lost protein functional features of individual exon skipping events in ROSMAP.
Lost protein functional features of individual exon skipping events in ROSMAP. Lost protein functional features of individual exon skipping events in MSBB.
Lost protein functional features of individual exon skipping events in MSBB. Lost protein functional features of individual exon skipping events in Mayo.
Lost protein functional features of individual exon skipping events in Mayo. 3'-UTR exon skipping evnets lost miRNA binding.
3'-UTR exon skipping evnets lost miRNA binding. - Differential PSIs between mutated versus non-mutated samples.
- Differential PSIs between mutated versus non-mutated samples. - Depth of Coverage in the skipped exon of the mutated samples.
- Depth of Coverage in the skipped exon of the mutated samples. - Sashimi plot in the skipped exon of the mutated samples.
- Sashimi plot in the skipped exon of the mutated samples. - Non-synonymous mutations located in the skipped exons.
- Non-synonymous mutations located in the skipped exons. - Non-synonymous mutations located in the skipped exons in CCLE.
- Non-synonymous mutations located in the skipped exons in CCLE. Associated exon skipping events with Braak staging or Clinical Dementia Rating (CDR).
Associated exon skipping events with Braak staging or Clinical Dementia Rating (CDR). sQTL information located at the skipped exons.
sQTL information located at the skipped exons. Correlated RBP and related information.
Correlated RBP and related information. Approved drugs targeting this gene.
Approved drugs targeting this gene.  Diseases associated with this gene.
Diseases associated with this gene. 